Beyond Lithium: A Battery Revolution?
Lithium-ion batteries power our phones, laptops, and cars. For decades, they've been the go-to for energy storage, offering a good balance of energy density and power. However, they have drawbacks: limited range, slow charging, safety concerns, and environmental issues from lithium mining are pushing the search for alternatives.
Graphene, a material known for its strength, lightness, and conductivity, promises faster charging, longer lifespans, and better safety in batteries. However, it's not a direct replacement for lithium-ion. Instead, graphene can enhance existing battery technology in several ways.
New battery technology typically evolves through gradual improvements rather than complete overhauls. Lithium-ion will remain relevant, but research into materials like graphene is vital for advancing possibilities. Graphene could become a valuable tool as energy storage demands increase.

Lithium-Ion: The Current Champion
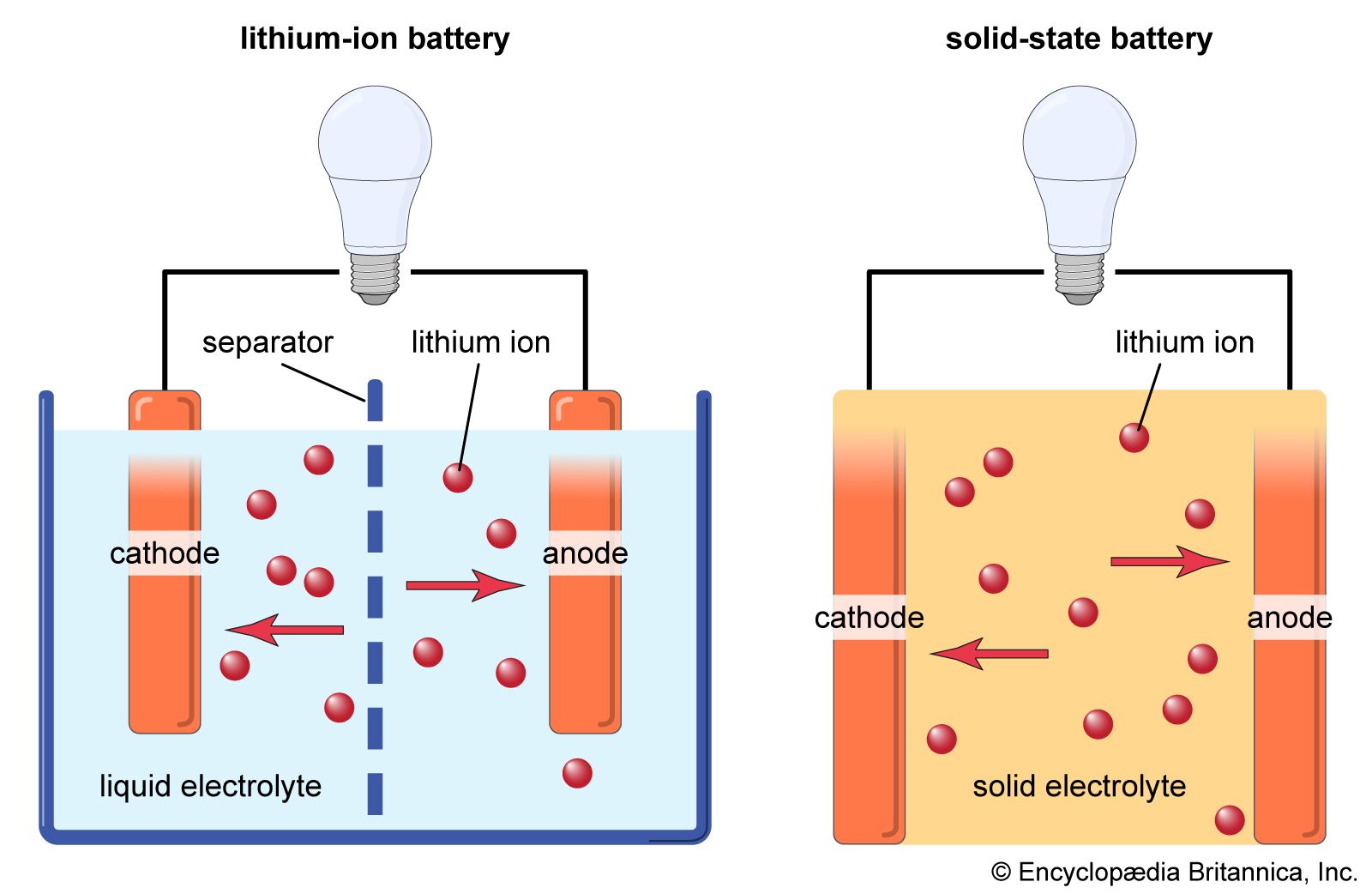
A lithium-ion battery has four main components: a cathode, an anode, an electrolyte, and a separator. The cathode and anode are electrodes for chemical reactions. The electrolyte facilitates ion movement, and the separator prevents short circuits. The materials in each part define the battery's performance.
Lithium-ion batteries come in various chemistries, each with trade-offs. NMC batteries, common in EVs, offer high energy density and good performance, but cobalt sourcing is an issue. NCA batteries are similar to NMC with higher energy density but potentially less stability. LFP batteries are safer, last longer, and cost less, but have lower energy density.
Lithium-ion's widespread use stems from years of refinement. They power everything from smartphones to electric vehicles and are used in grid storage to balance renewable energy. Established manufacturing and a mature supply chain also support their adoption.
Lithium-ion batteries have drawbacks. They can experience thermal runaway, causing fires. Performance degrades over time, and energy density limits some applications. Lithium mining's environmental impact, including water use and habitat disruption, is also a growing concern, fueling the search for alternatives like graphene.
- NMC: High energy density, good performance, cobalt sourcing concerns.
- NCA: Even higher energy density, potentially less stable.
- LFP: Safe, long lifespan, lower cost, lower energy density.
Lithium-Ion Chemistry Comparison (Projected for 2026)
| Chemistry | Typical Applications | Key Advantages | Potential Drawbacks | Temperature Performance |
|---|---|---|---|---|
| NMC (Nickel Manganese Cobalt) | Electric Vehicles, Power Tools, Laptops | Offers a good balance of performance characteristics | Cobalt sourcing can be ethically complex | Moderate sensitivity to extreme temperatures |
| NCA (Nickel Cobalt Aluminum) | High-Performance EVs, Grid Storage | Generally provides high energy density for longer range | Can be more prone to thermal runaway if not properly managed | Requires sophisticated thermal management systems |
| LFP (Lithium Iron Phosphate) | Energy Storage Systems, Buses, Lower-Cost EVs | Known for its enhanced thermal and chemical stability | Typically has lower energy density than NMC or NCA | Good performance across a wider temperature range |
| Graphene-Enhanced Lithium-Ion | Emerging applications in various sectors | Potential for faster charging and improved lifespan | Manufacturing scalability and cost remain significant hurdles | May exhibit improved temperature tolerance depending on implementation |
| Solid-State Lithium-Ion (with Graphene) | Next-generation EVs, High-density portable devices | Promises increased safety and higher energy density | Still in early stages of development and faces production challenges | Expected to have improved thermal stability |
Qualitative comparison based on the article research brief. Confirm current product details in the official docs before making implementation choices.
Graphene's Promise: What Makes It Different?
Graphene is a single layer of carbon atoms in a hexagonal lattice, one atom thick. It's about 200 times stronger than steel and possesses high electrical and thermal conductivity, along with flexibility. These properties make it attractive for batteries.
Graphene is not a battery type but a material that enhances existing battery technologies. It can be used as an electrode material (replacing or augmenting graphite), a conductive additive to improve electron transport, or in separators to enhance ion transport and prevent dendrite formation.
Using graphene as a conductive additive is a near-term application. Small amounts can significantly improve electrode conductivity, enabling faster charging and discharging. As an electrode material, graphene's large surface area can increase energy density by providing more sites for lithium ion interaction. In separators, it can create a more stable and efficient barrier.
Graphene Battery Types: Beyond the Hype
Graphene-based batteries are being developed in several ways, with varying maturity. Graphene-enhanced lithium-ion batteries are the most common and closest to market. They use graphene as an additive to improve existing lithium-ion performance, offering incremental gains in charging speed, lifespan, and conductivity without major manufacturing changes.
Graphene-sulfur batteries are more ambitious. Sulfur is cheap and abundant but has poor conductivity and low energy density. Graphene can act as a scaffold to improve conductivity and allow more sulfur use. These batteries could offer higher energy density than lithium-ion but face cycle life and stability challenges.
Fully graphene batteries, with graphene anodes and cathodes, are mostly in research. They often use different electrolytes and operating mechanisms than lithium-ion. While they could offer very high performance, challenges in material synthesis, scalability, and cost remain. Commercial availability is distant.
Focusing on current developments, graphene-enhanced lithium-ion batteries are appearing in consumer electronics and EVs, offering modest improvements. Other graphene battery technologies are still under development with uncertain commercial viability.
Performance Face-Off: Graphene vs Lithium-Ion
Comparing graphene-based batteries to lithium-ion is complex, as graphene isn't a single technology and performance varies with integration. For energy density, graphene-enhanced lithium-ion offers some improvement but doesn't yet match the theoretical potential of graphene-sulfur or fully graphene batteries. Commercially available lithium-ion generally leads in energy density.
Charging speed is where graphene truly shines. Graphene’s high conductivity allows for much faster electron transport, enabling significantly faster charging times. Graphene-enhanced batteries can often be charged in a fraction of the time it takes to charge a comparable lithium-ion battery. Lifespan is another area where graphene can provide an advantage, improving the cycle life of batteries and reducing degradation over time.
Safety is a major concern with lithium-ion batteries, and graphene can help to mitigate this risk. Graphene’s thermal stability and ability to prevent dendrite formation can reduce the likelihood of thermal runaway. Cost is a significant barrier to graphene adoption. Graphene production is still relatively expensive, although prices are falling as manufacturing processes improve. Environmental impact is complex. While lithium mining has its own issues, the environmental impact of large-scale graphene production is still being studied.
It is important to note that many of the performance claims surrounding graphene batteries are based on laboratory results and haven’t been fully validated in real-world applications. Direct comparison is difficult because graphene battery development is constantly evolving. As of late 2023, commercially available graphene-enhanced batteries offer incremental improvements, while the more revolutionary graphene-based technologies are still in the research and development phase.
Featured Products

20000mAh capacity with 45W fast charging · Built-in cable for convenience · 3 ports for multiple device charging
This graphene-enhanced power bank offers a glimpse into the future of portable charging, showcasing the potential benefits of this advanced battery technology for consumers.

10000mAh capacity with 45W PD fast charging · Ultra-compact design · Built-in USB-C cable
This graphene-enhanced power bank offers a glimpse into the future of portable charging, showcasing the potential benefits of this advanced battery technology for consumers.

15000mAh capacity with 22.5W fast charging · Built-in cable for convenience · 6 output ports
This graphene-enhanced power bank offers a glimpse into the future of portable charging, showcasing the potential benefits of this advanced battery technology for consumers.

20000mAh capacity with 87W max fast charging · Built-in USB-C cable · 3 ports for versatile charging
This graphene-enhanced power bank offers a glimpse into the future of portable charging, showcasing the potential benefits of this advanced battery technology for consumers.

10000mAh capacity with 45W fast charging · Ultra-mini size for portability · Built-in USB-C cable
This graphene-enhanced power bank offers a glimpse into the future of portable charging, showcasing the potential benefits of this advanced battery technology for consumers.
As an Amazon Associate I earn from qualifying purchases. Prices may vary.
2026 Predictions: Where Will We Be?
By 2026, I don’t anticipate graphene batteries completely replacing lithium-ion. The established infrastructure and economies of scale for lithium-ion are too significant to overcome quickly. However, I expect to see a much wider adoption of graphene-enhanced lithium-ion batteries, particularly in consumer electronics, electric scooters, and potentially some electric vehicles.
Niche applications where fast charging and long lifespan are paramount – such as high-performance drones and power tools – are also likely to see increased graphene battery adoption. We might also see graphene-enhanced batteries used in grid storage applications to improve efficiency and reliability. Fully graphene batteries and graphene-sulfur batteries are likely to remain in the research and development phase, with limited commercial availability.
The success of graphene batteries will depend on several factors. Reducing the cost of graphene production is crucial. Scaling up manufacturing processes to meet demand will also be a challenge. Overcoming regulatory hurdles and establishing clear safety standards are essential. And, of course, continued research and development are needed to unlock the full potential of graphene battery technology.
The Supply Chain Question
The sourcing of materials for both lithium-ion and graphene batteries raises important ethical and environmental concerns. Lithium mining, particularly in South America’s “Lithium Triangle,” can have significant impacts on water resources and local ecosystems. The extraction process can also contribute to soil contamination and habitat destruction. Cobalt, another key component of many lithium-ion batteries, is often mined in the Democratic Republic of Congo, where labor practices are often exploitative.
Graphene production, while potentially more sustainable, isn’t without its challenges. The most common method of graphene production involves using graphite, which is typically sourced from China. However, research is underway to develop more sustainable methods of graphene production, such as using biomass or carbon dioxide as feedstock. These methods could significantly reduce the environmental impact of graphene production.
Ultimately, the most sustainable battery technology will be the one that minimizes environmental impact throughout its entire lifecycle, from material sourcing to manufacturing to end-of-life recycling. Both lithium-ion and graphene batteries have their own strengths and weaknesses in this regard, and ongoing research is needed to address these challenges.
Beyond Batteries: Graphene’s Wider Impact
The potential of graphene extends far beyond batteries. Its exceptional properties make it a promising material for a wide range of applications, including high-speed transistors for electronics, strong and lightweight composites for aerospace and automotive industries, and highly sensitive sensors for medical diagnostics and environmental monitoring.
The continued investment in graphene research and development, driven in part by the demand for better batteries, will undoubtedly lead to breakthroughs in these other areas as well. It’s a versatile material with the potential to revolutionize numerous industries, and batteries are just one piece of the puzzle.





No comments yet. Be the first to share your thoughts!